 In this way energy in food molecules (like glucose) are released gradually instead of big bursts. Together, these reactions comprise cellular respiration.Įnergy in cells is generated by moving electrons from one chemical to another. The resulting change in free energy is used to synthesize ATP from ADP and P. Living organism can now use this energy to do work. In cells, glucose is oxidized through a long series of carefully controlled redox reactions. In this way energy is released from the glucose in small amounts. And oxygen becomes reduced, because it gains electrons. Each carbon atom loses electrons when it is oxidized in the presence of oxygen. Glucose goes through the same type of reaction. Hence, these reactions are known as redox reactions. And this is how living things generate usable energy from energy stored in molecules. For every reduction, there is an oxidation. A simple redox reaction is the ionic bonding of salt (NaCl), where a sodium loses an electron to chlorine generating a positive charged sodium and a negatively charged chlorine. By gaining electrons, it creates a positive charge on that atom or electron. In contrast, Reduction is the gain of electrons by a molecule or atom. All eukaryotic organisms can convert use glucose as food. In general, aerobic respiration is the breakdown of food in the presence of oxygen producing carbon dioxide, water and synthesizing ATP. 
General Equation for Cellular Respiration 
This displacement from equilibrium means that the hydrolysis of ATP in the cell releases a large amount of free energy. Living cells maintain the ratio of ATP to ADP at a point ten orders of magnitude from equilibrium, with ATP concentrations a thousandfold higher than the concentration of ADP. A system that is far from equilibrium is capable of doing work. Thus, if ATP and ADP are in chemical equilibrium in water, almost all of the ATP will eventually be converted to ADP. This is because the strength of the bonds between the phosphate groups in ATP are less than the strength of the hydrogen bonds, between its products (ADP + phosphate), and water. ATP is an unstable molecule in water, in which it hydrolyses to ADP and phosphate. ATP is then split into ADP (which is now relaxed) and a phosphate molecule.ĪTP consists of adenosine and three phosphate groups (triphosphate). It is a lot like a chemical spring that gets loaded, and move around a cell, and can be split apart producing energy. ATPĪdenosine Triphosphate is commonly known as ATP. Through cellular respiration and fermentation, those bonds are broken releasing the potential energy of organic molecules into kinetic energy that cells use to do work. Cells release the energy in those organic molecules by breaking them down. For nearly all organisms on Earth (except chemolithotrophs), that energy is stored in organic molecules. 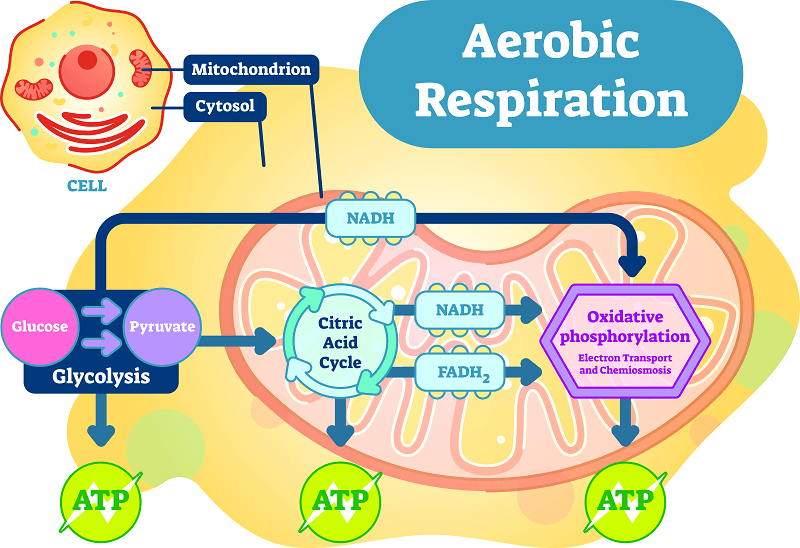
Any chemical process that yields energy is known as a catabolic pathway. 
The energy stored in ATP (its third phosphate group is weakly bonded to the rest of the molecule and is cheaply broken allowing stronger bonds to form, thereby transferring energy for use by the cell) can then be used to drive processes requiring energy, including biosynthesis, locomotion or transportation of molecules across cell membranes.Ĭellular respiration and fermentation produce energy for cells to use. Nutrients that are commonly used by animal and plant cells in respiration include sugar, amino acids and fatty acids, and a common oxidizing agent (electron acceptor) is molecular oxygen (O2). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
