 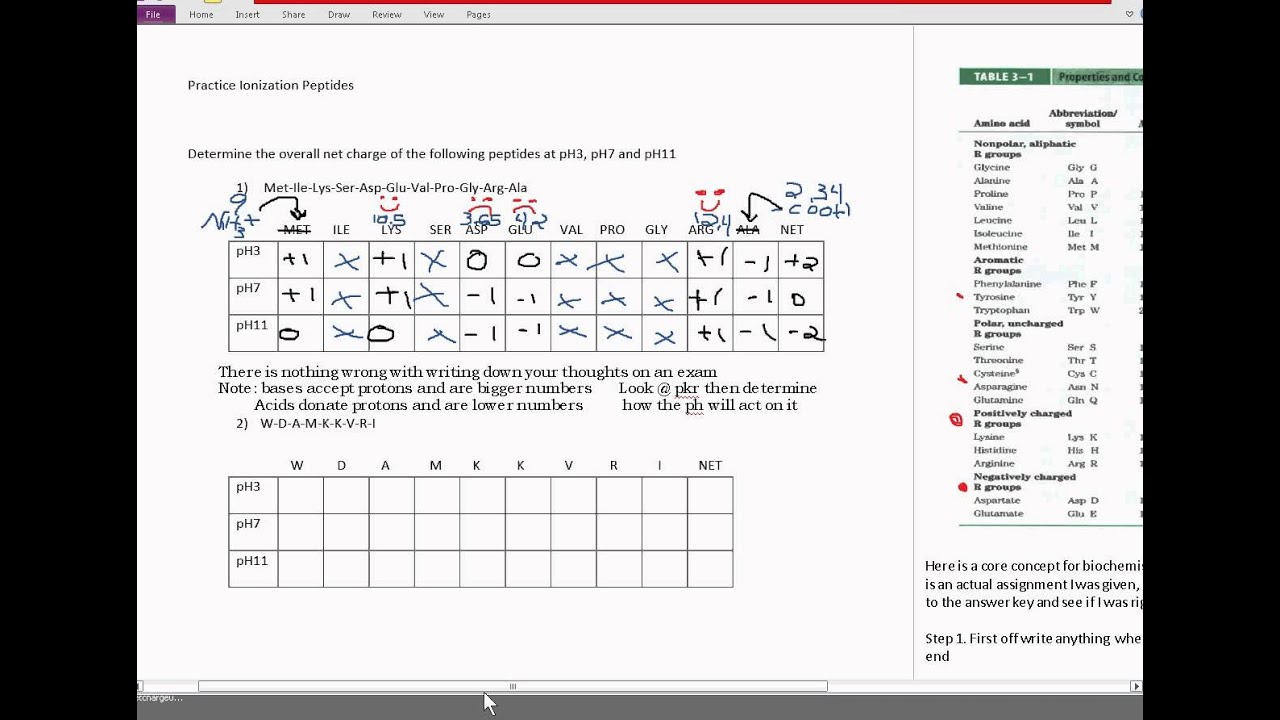
Similarly, detergents such as SDS (sodium dodecyl sulfate, also called sodium lauryl sulfate) can solubilize proteins, resulting in a hydrophobic core that promotes denaturation of the protein. They can even overcome the hydrogen bonds and other side chain interactions that hold α-helices and β-pleated sheets intact. They can disrupt tertiary and quaternary structures by breaking disulfide bridges, reducing cystine back to two cysteine residues. On the other hand, solutes such as urea denature proteins by directly interfering with the forces that hold the protein together. This is what happens when egg whites are cooked: In the uncooked egg whites, albumin is folded, which makes it transparent cooking them causes the albumin molecules to denature and aggregate, forming a solid, rubbery white mass that will not revert to its transparent form. When the temperature gets high enough, this extra energy can be enough to overcome the hydrophobic interactions that hold a protein together, causing the protein to unfold. Finally, when we've added 2.0 equivalents of base, the amino acid has become fully deprotonated, and all that remains is NH CH COO additional base will only increase the pH further.Īs with all molecules, when the temperature of a protein increases, its average kinetic energy increases. Once again, the titration curve is nearly horizontal. When 1.5 equivalents of base have been added, the concentration of the zwitterion form equals the concentration of the fully deprotonated form that is, =, and the pH equals pK. The peptide molecular weight calculator will display average and monoisotopic mass of the molecule, as well as a table of mass divided by charge values, both in positive and negative scan modes, which is useful for mass spec analysis. For neutral amino acids, it can be calculated by averaging the two pK values for the carboxylic acid and amino groups: As we continue adding base, glycine passes through a second buffering phase as the amino group deprotonates again, the pH remains relatively constant. Simply type in, or copy and paste, peptide or protein fragment amino-acid sequence, including modifications, spacers, or special termini, and press the Calculate button. This is true of all amino acids: the isoelectric point is the pH at which the molecule is electrically neutral. This means that every molecule is now electrically neutral, and thus the pH equals the isoelectric point (pI) of glycine. When we've added 1.0 equivalent of base, glycine exists exclusively as the zwitterion form (remember, we started with 1.0 equivalent of glycine). The amino acid stops acting like a buffer, and pH starts to increase rapidly during this phase. Remember: when the pH is close to the pK value of a solute, a solution is acting as a buffer, and the titration curve is relatively flat, as demonstrated in the blue box in the diagram.As we add more base, the carboxylate group goes from half-deprotonated to fully deprotonated. When 0.5 equivalents of base have been added to the solution, the concentrations of the fully protonated glycine and its zwitterion, +NH3CH2COO-, are equal that is, =. As the solution is titrated with NaOH, the carboxyl group will deprotonate first because it is more acidic than the amino group. 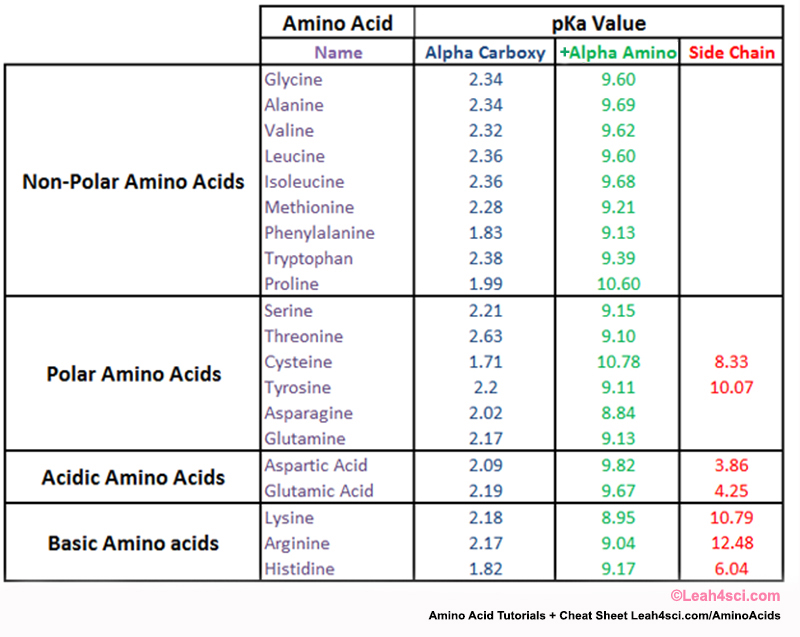
At low pH values, glycine exists predominantly as NH3CH2COOH it is fully protonated, with a positive charge. The two proteins are not highly conserved.Imagine an acidic 1 M glycine solution. testosteroni isomerase lacks these amino acids. putida isomerase contains 3 cysteines and 2 tryptophans, whereas the P. Comparison of the two proteins shows that the region of strongest homology is the region containing the aspartic acid at which steroidal affinity and photoaffinity reagents have been shown to react in the P. 
Only the oxosteroid isomerase from Pseudomonas testosteroni, an expected homology, was found to be similar. A search for homologous proteins was performed. The full sequence was deduced by the overlapping of the various peptides. The peptides resulting from each fragmentation were separated by reversed-phase high performance liquid chromatography and sequenced by automated Edman degradation. The isomerase was fragmented using CNBr, trypsin, iodosobenzoic acid, and acid cleavage at aspartyl-prolyl peptide bonds. The intact S-carboxymethyl protein was sequenced from the NH2 terminus using standard automated Edman degradation and automated Edman degradation using fluorescamine treatment at known prolines to suppress background. The enzyme is a dimeric protein of two identical subunits, each consisting of a polypeptide chain of 131 residues and a Mr = 14,536. We have determined the primary structure of a delta 5-3-oxosteroid isomerase from Pseudomonas putida biotype B. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
